Fanapt® significantly improved overall schizophrenia
symptoms as measured by PANSS1,2
Improvement in symptoms in a 4-week study2

-
In this study, Fanapt® (iloperidone) demonstrated
similar efficacy to ziprasidone, which also required a slow
titration to the target dose1
Study design: 4-week, placebo- and
active-controlled trial (n=604) involved one fixed dose of
Fanapt® (24 mg/day) compared to placebo- and an
active-control (ziprasidone). The titration was similar to that of
the 6-week study. Fanapt® was titrating at 1 mg twice
daily on Day 1, and the dose was increased to 2, 4, 6, 8, 10, and 12
mg twice daily on Days 2, 3, 4, 5, 6, and 7, respectively. The
primary endpoint was change from baseline on the PANSS total score
at the end of treatment (Day 28) analyzed using mixed-effects model
repeated measures (MMRM). Mean baseline scores were 92.88 for
Fanapt® and 90.48 for placebo.1,3
PANSS-T = Positive and Negative Syndrome Scale total score.
Fanapt® significantly improved overall schizophrenia
symptoms as measured by BPRS1,3
Improvement in symptoms in a 6-week study3
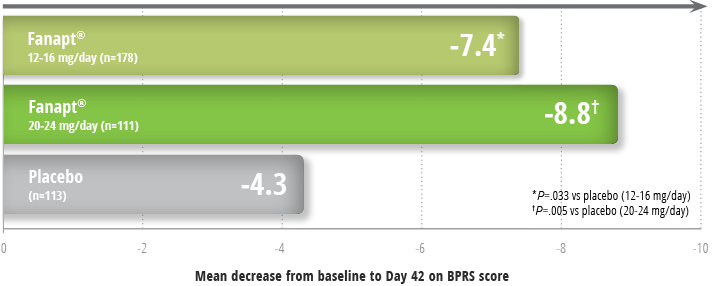
Study design: 6-week, placebo- and
active-controlled trial (n=706) involving 2 flexible dose ranges of
Fanapt® (12 to 16 mg/day or 20 to 24 mg/day) compared to
placebo and an active-control (risperidone). Fanapt® was
titrated to the respective dose over a period of 7 days. The primary
endpoint was change from baseline on the BPRS total score at the end
of treatment (Day 42).1
Patients taking Fanapt® at both high and low doses
demonstrated significant reductions from baseline in BPRS score at
Day 421:
-
The active-control antipsychotic drug (risperidone) appeared to be
superior to Fanapt® in this trial within the first 2
weeks, a finding that may in part be explained by the more rapid
titration that was possible for that drug. In patients in this
study who remained on treatment for at least 2 weeks, iloperidone
appeared to have had comparable efficacy to the active control
(risperidone)
BPRS = Brief Psychiatric Rating Scale; LOCF = Last observation
carried forward.
Fanapt® demonstrated efficacy with placebo-like rates
of akathisia.1
Find out more
References: 1. Fanapt [package
insert]. Washington, DC: Vanda Pharmaceuticals, Inc.: 2016.
2. Cutler AJ, Kalahi AH, Weiden PJ, Hamilton J,
Wolfgang CD. Four-week, double-blind, placebo- and
ziprasidone-controlled trial of iloperidone in patients with acute
exacerbations of schizophrenia. J Clin Psychopharmacol.
2008;28(2 Suppl 1):S20-S28. 3. Data on File, Vanda
Pharmaceuticals, Inc.
RELAPSE PREVENTION
FANAPT® DEMONSTRATED LONG-TERM EFFICACY BY DELAYING
TIME TO RELAPSE
FANAPT® DEMONSTRATED EFFICACY FOR LONG-TERM MAINTENANCE
TREATMENT OF SCHIZOPHRENIA
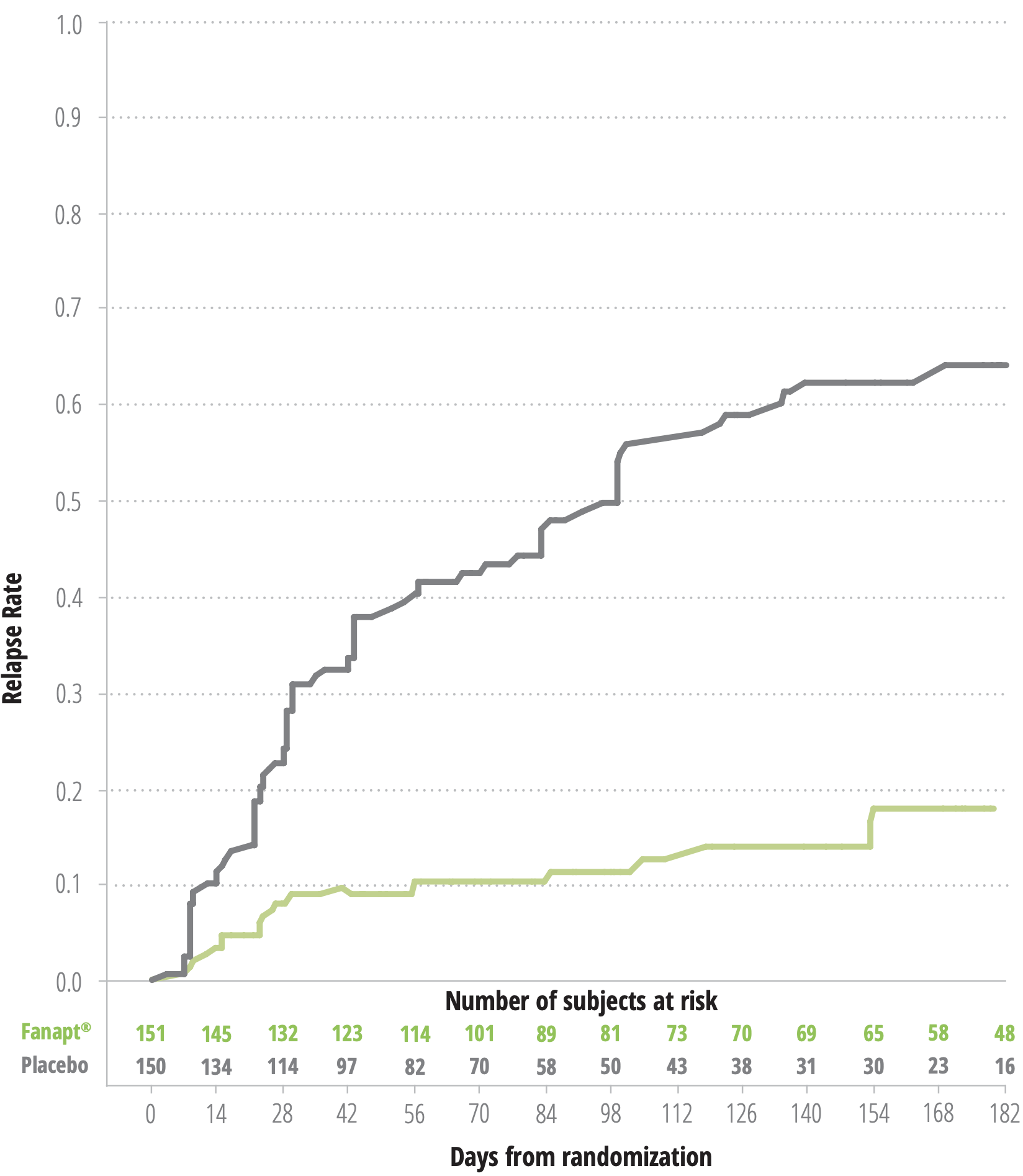
KAPLAN MEIER ESTIMATION OF PERCENT RELAPSE/IMPENDING RELAPSE FOR
FANAPT® AND PLACEBO-TREATED PATIENTS1*
(log rank test: P<.0001)
*Based on final analysis dataset.
-
BASED ON THE INTERIM ANALYSIS, THE MEAN TIME TO RELAPSE WAS 139
DAYS FOR FANAPT® VS 71 DAYS FOR PLACEBO4*
-
AFTER 6 MONTHS, 79.6% OF PATIENTS TAKING FANAPT® DID NOT
RELAPSE VS 36.6% ON PLACEBO, BASED ON THE INTERIM ANALYSIS4
-
NO NEW SAFETY SIGNALS COMPARED TO THOSE OBSERVED IN SHORT-TERM
STLIDIES4
-
FLEXIBLE DOSING OF FANAPT® (8-24 MG/DAY) WAS EFFECTIVE1
*Relapse or impending relapse is defined as any
of the following: hospitalization due to worsening of
schizophrenia; increase
(worsening) of the PANSS score ≥30%; CGI-improvement score of
≥6; patient had suicidal, homicidal, or aggressive behavior;
CGI, Clinical Global Impression.
IMPORTANT SAFETY INFORMATION
Neuroleptic malignant syndrome, a
potentially fatal symptom, has been reported in association with
antipsychotic drugs, including Fanapt®. Manage with immediate
discontinuation of drug, treatment if needed, and close
monitoring.
Tardive dyskinesia: The risk of tardive
dyskinesia may increase as the duration of treatment and total
cumulative dose increases. Discontinue Fanapt® if
clinically appropriate.
Seizures: Use Fanapt®
cautiously in patients with a history of seizures or with
conditions that lower seizure threshold.
Falls: Fanapt® may cause
somnolence, postural hypotension, motor and sensory instability,
which may lead to falls causing fractures or other injuries. For
patients with diseases, conditions or medications that could
exacerbate these effects, complete fall risk assessments initially
and recurrently during therapy.
Leukopenia, neutropenia, and agranulocytosis
have been reported with antipsychotics. Patients with a
pre-existing low white blood cell count (WBC) or a history of
leukopenia/neutropenia should have their complete blood count
(CBC) monitored frequently during the first few months of therapy
and should discontinue Fanapt at the first sign of a decline in
WBC in the absence of other causative factors.
Study design: A double-blind, placebo-controlled,
randomized withdrawl study (n=303) involving a flexible-dose range
of Fanapt® (8-24 mg/day) administered as twice-daily
doses. The study consisted of a 7-day titration period where
Fanapt® was titrated at 1 mg twice daily on day 1,2 mg
twice daily on day 2,4 mg twice daily on day 3,6 mg twice daily on
day 4, 5, 6 and 7, followed by a stabilization period lasting at
least 12 weeks. During the subsequent double-blind relapse
prevention phase lasting up to 26 weeks, patients were randomized
to placebo or established dose of Fanapt® and observed
for relapse or impending relapse. Stablization was defined as
being on an established dose of Fanapt® that was
unchanged due to efficacy in the 4 weeks prior to randomization,
having a CGI-Severity score ≤4 and PANSS score ≤70, a score
of ≤4 on each of the following individual PANSS items
(P1-delusions, P2-conceptual disorganization, P3-hallucinatory
behavior, P6-suspiciousness/persecution, P7-hostility, or G8-
uncooperativeness), and no hospitalization or increase in level of
care to treat exacerbations. The primary endpoint was time to
relapse or impending relapse compared to placebo, defined as any
of the following: hospitalization due to worsening of
schizophrenia; increase (worsening) of PANSS score of ≥30%;
CGI-Improvement score ≥6; patient had suicidal, homicidal, or
aggressive behavior; or need for any other antipsychotic
medication, including increase in study medication.